Environmental Laboratory
Certified Reference Microorganisms
Feb 27 2018
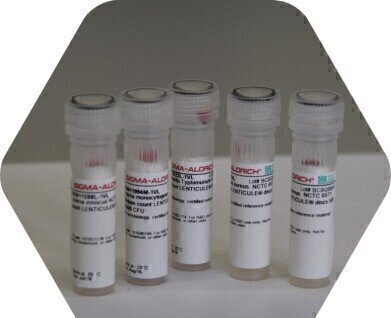
Vitroids™ and LENTICULE® discs contain viable microorganisms in a certified quantity (ISO 17025), produced under reproducible conditions (ISO Guide 34) with strains obtained from CECT®, NCTC and other collections. The discs consist of bacteria or fungi in a solid water-soluble matrix. Microorganisms in this form are stable for at least one year and are in a viable stage (no lag phase or recovery time). Merck provides two product lines of microbiology CRMs. The LENTICULE® discs are produced in partnership and under license from PHE with strains from NCTC and NCPF. The Vitroids™ are Merck′s own innovative development of discs containing CECT® strains. These discs contain defined certified colony forming units (CFUs) with a standard deviation and an expanded uncertainty. The product concentrations are designed in a range where no or only minimal dilution must be done. The discs dissolve in liquid media and on agar plates which mean easy handling and very economical solution.
Digital Edition
IET 34.2 March 2024
April 2024
Gas Detection - Biogas batch fermentation system for laboratory use with automatic gas analysis in real time Water/Wastewater - Upcycling sensors for sustainable nature management - Prist...
View all digital editions
Events
Apr 30 2024 Melbourne, Australia
Apr 30 2024 Birmingham, UK
May 03 2024 Seoul, South Korea
May 05 2024 Seville, Spain
May 06 2024 Minneapolis, MN, USA